H-Bonding
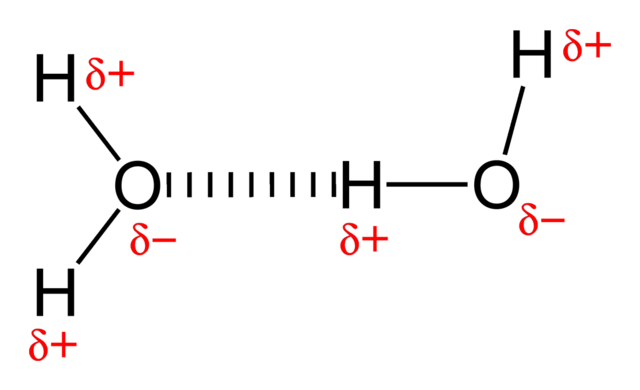
H-Bonding is a force that will be present in this molecule. The hydrogen will be able to bond with the oxygen of the Glucose, and the hydrogen of the glucose can bond to the O, N, or F of any other molecule that has it. The diagram to the left does not show glucose specifically, but it shows the force that will happen when it comes across another H, N, O, or F.
Dipole - Dipole
Dipole-Dipole can occur with this molecule. The positive end of this molecule can make a bond with the megative side of another polar molecule, and vise-versa. This cannot happen with non-polar molecules like CCl4.
Dispersion
Dispersion is present in this molecule because it happens between every two molecules. Although it is very short and very weak, it will happen momentarily between every two molecules.
